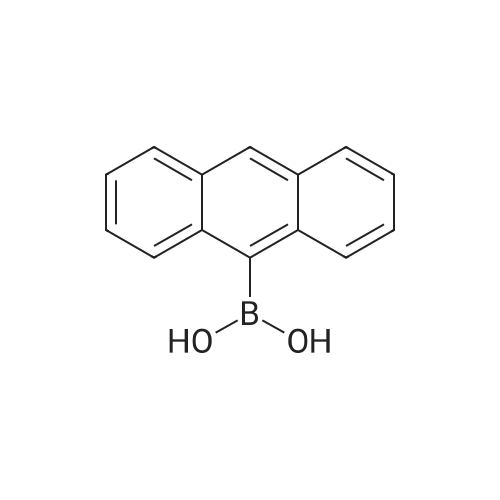
ChemSpider SyntheticPages | Stille coupling of thiophene to 1,2,4,5-tetrabromo-3,6-bis(dodecyloxy)benzene

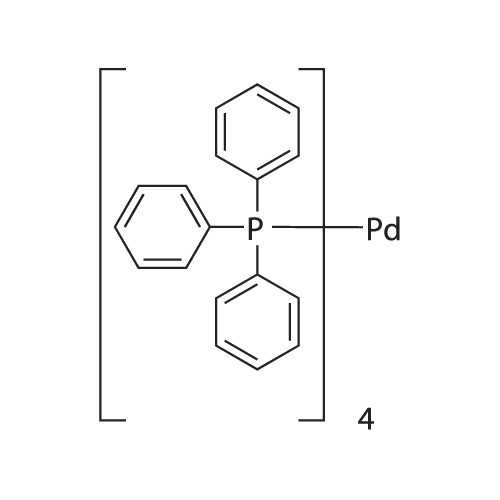
Palladium Acetate Trimer: Understanding Its Ligand-Induced Dissociation Thermochemistry Using Isothermal Titration Calorimetry, X-ray Absorption Fine Structure, and 31P Nuclear Magnetic Resonance | Organometallics

Halogen‐Bridged Methylnaphthyl Palladium Dimers as Versatile Catalyst Precursors in Coupling Reactions - Sivendran - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Linear α-Olefins Obtained with Palladium(II) Complexes Bearing a Partially Oxidized Tetraphosphane | Organometallics

Can Palladium Acetate Lose Its “Saltiness”? Catalytic Activities of the Impurities in Palladium Acetate | Organic Letters
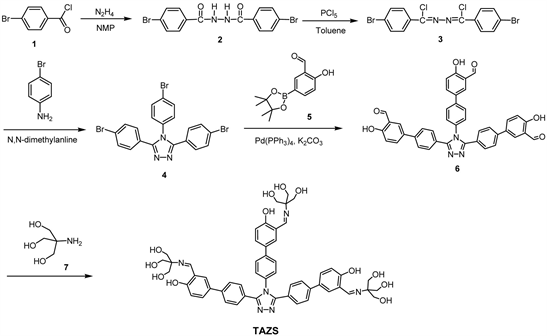
Solution Processable Material Derived from Aromatic Triazole, Azomethine and Tris: Preparation and Hole-Buffering Application in Polymer Light-Emitting Diodes

Palladium Acetate Trimer: Understanding Its Ligand-Induced Dissociation Thermochemistry Using Isothermal Titration Calorimetry, X-ray Absorption Fine Structure, and 31P Nuclear Magnetic Resonance | Organometallics

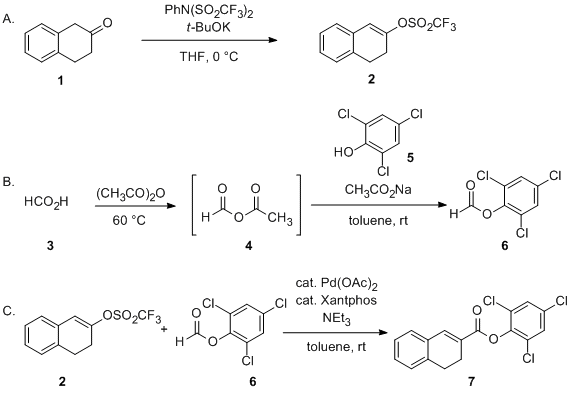
Palladium‐Catalyzed Asymmetric Hydrogenolysis of Aryl Triflates for Construction of Axially Chiral Biaryls - Li - 2023 - Angewandte Chemie International Edition - Wiley Online Library

Use of Polymer-Supported Dialkylphosphinobiphenyl Ligands for Palladium-Catalyzed Amination and Suzuki Reactions | The Journal of Organic Chemistry

Palladium‐Catalyzed Directed Atroposelective C−H Iodination to Synthesize Axial Chiral Biaryl N‐Oxides via Enantioselective Desymmetrization Strategy - Yao - 2023 - Chemistry – A European Journal - Wiley Online Library

Can Palladium Acetate Lose Its “Saltiness”? Catalytic Activities of the Impurities in Palladium Acetate | Organic Letters

Palladium Acetate Trimer: Understanding Its Ligand-Induced Dissociation Thermochemistry Using Isothermal Titration Calorimetry, X-ray Absorption Fine Structure, and 31P Nuclear Magnetic Resonance | Organometallics

Can Palladium Acetate Lose Its “Saltiness”? Catalytic Activities of the Impurities in Palladium Acetate | Organic Letters

Synthesis and Structures of Mono- and Trinuclear Complexes of Palladium(II) with 2-Chloropyridine | SpringerLink

Can Palladium Acetate Lose Its “Saltiness”? Catalytic Activities of the Impurities in Palladium Acetate | Organic Letters
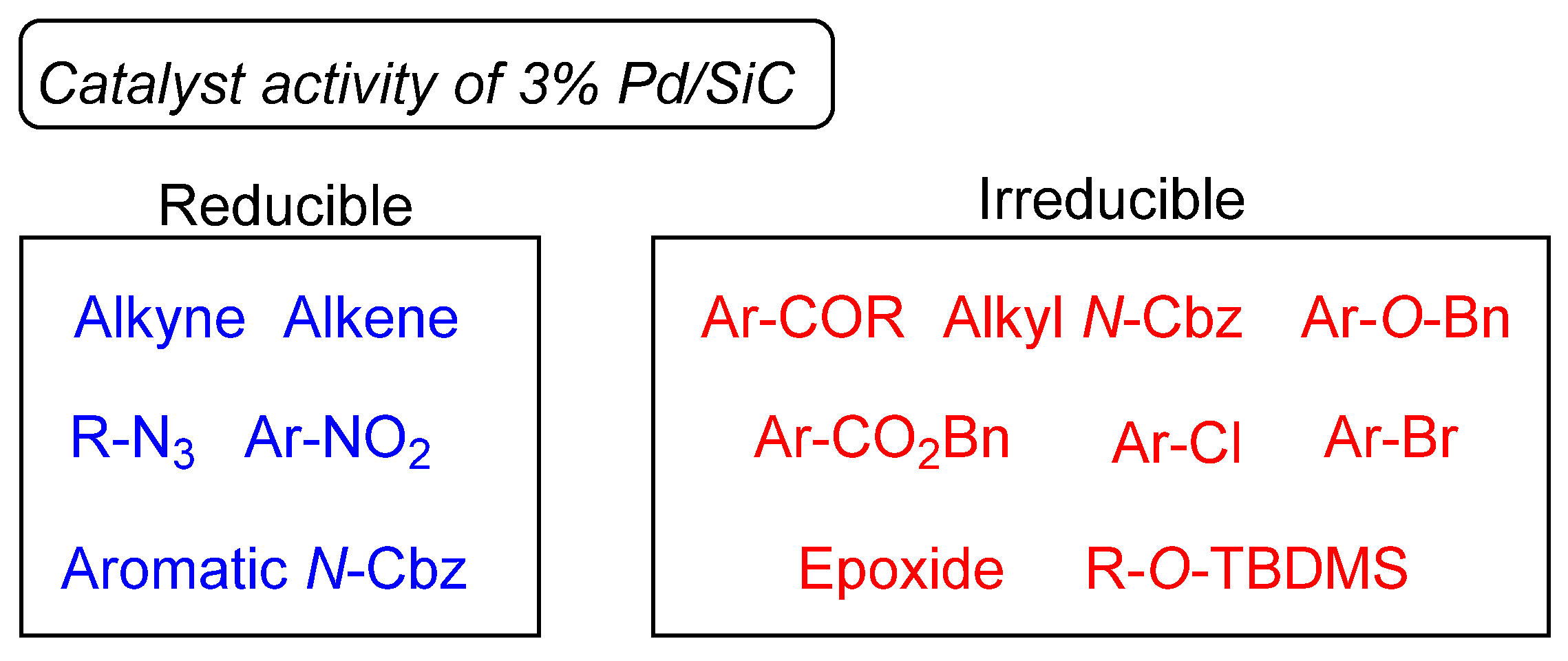
Catalysts | Free Full-Text | Development of Silicon Carbide-Supported Palladium Catalysts and Their Application as Semihydrogenation Catalysts for Alkynes under Batch- and Continuous-Flow Conditions